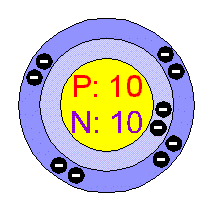
Neon has its outer electrons removed from a 2p orbital rather than. Similarly, the atoms of all group 7 elements have similar chemical properties and reactions to each other because all of them have seven electrons in their outer shell. Explain why the value of the first ionisation energy of neon is higher than that of sodium. The atoms of all group 1 elements have similar chemical properties and reactions because they all have one electron in their outer shell. The electronic structures of the elements in group 1: 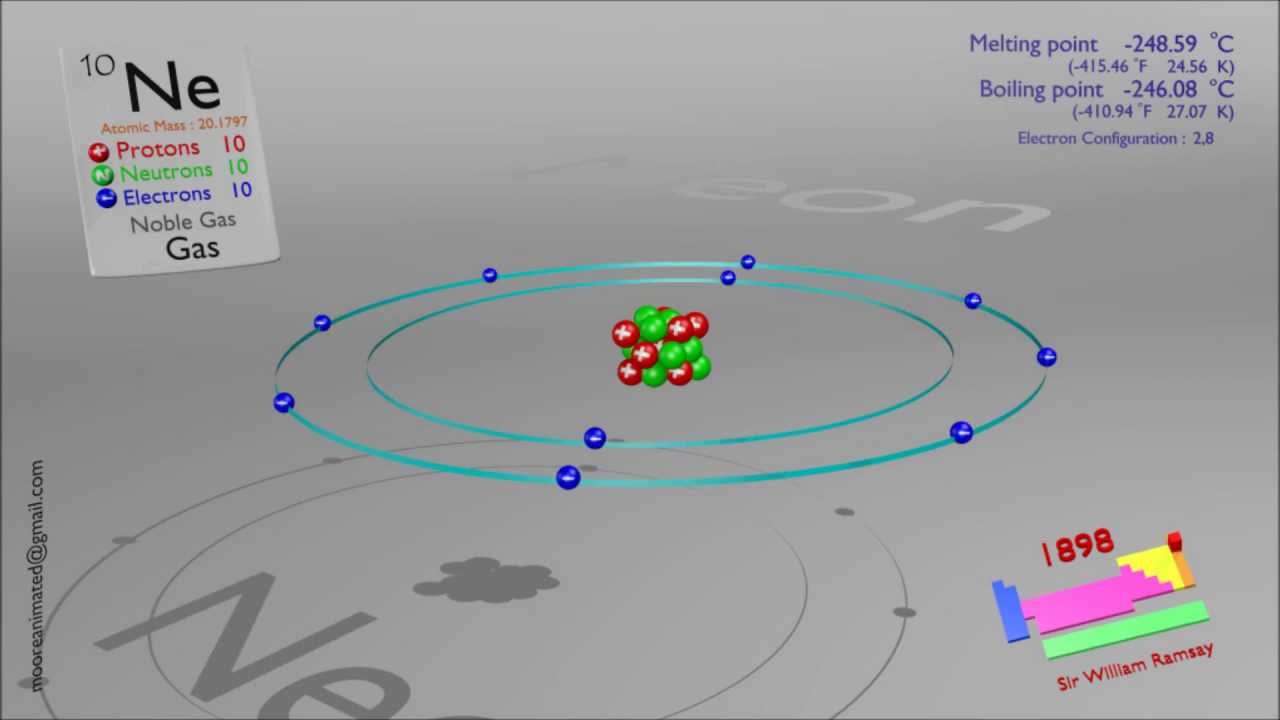
So, elements in the same group have similar chemical properties because they have the same number of electrons in their outer shell. When atoms collide and react, it is the outer electrons that meet and interact. Particles and atomic structure Revise 1 2 3 4 5 Structured questions This is the most common question on exam papers, although the number of marks for each question may vary. The electronic structures of atoms help explain the properties of elements and the structure of the periodic table. These elements have full outer shells - helium is 2, neon is 2.8, argon is 2.8.8, and their group number is group 0Įlectronic structures and properties of elements Browse royalty-free stock vectors of Atomic Structure yellow glowing neon icon and use them in your creative projects. It is less expensive refrigerant than helium in many applications. Helium, neon and other elements in this group In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. Table of contents What Is Neon Recommended Video Of Neon Chemical Properties Of Neon Uses Of Neon FAQs What is Neon Neon is reddish-orange coloured in neon lamps and vacuum discharge tubes and the second-lightest noble gas. Has only 1 electron but not placed in group 1 as it does not share properties with the rest of the group - it is placed by itself on the periodic table Note that there are some exceptions to the pattern shown with the example above. has an atomic number of (2 + 8 + 1) = 11 Figure 1 shows an overhead view of an atomic model completed by a student to show the proton and electron configuration of neon (neutrons are omitted for.The electronic structure of sodium (2.8.1) shows that sodium, Na: The electronic structure of an element is linked to its position on the periodic table. Find neon atomic structure Stock Photos & Images at agefotostock, one of the best stock photography sites. There is a maximum of seven shells, which can hold a different number of electrons, the electrons assignet to the outermost shell are known as valence electrons.Electronic structures and the periodic table The next heavier atom, the noble gas helium, consists of two electrons, two protons, and two neutrons.Īccording to the Bohr model of the atom the electrons are assigned to so-called shells, which represent different energy levels and therefore are arranged concentrically around the nucleus. The simplest atom is the hydrogen atom, with only one electron, one proton, and no neutron. A naturally occurring atom is electrically neutral, there are just as many positive protons as negatively charged electrons inside an atom. 
The positively charged protons and the neutrons form the nucleus, which is encircled by the electrons in certain intervals. 
Depending on the atom, it is composed of a certain number of electrons, protons, and neutrons. An atom is the smallest chemically not further divisible component of matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
